Defects such as porosity, oxide inclusions, and inconsistent mechanical properties are almost always linked to poor melt treatment. One of the most effective yet still underestimated tools for controlling melt quality is refining flux for aluminum.
When properly selected and correctly applied, refining flux improves melt cleanliness, stabilizes production, reduces scrap rates, and significantly enhances casting consistency.
This guide explains how refining flux actually works in industrial environments, how to apply it correctly, and why it remains essential in modern aluminum foundries.
What Is Refining Flux for Aluminum and Why Is It Important in Casting Production?
Refining flux for aluminum is a chloride- and fluoride-based chemical mixture designed to purify molten aluminum by removing unwanted gases and non-metallic impurities.
The main contaminants it targets include:
- Dissolved hydrogen (major cause of porosity)
- Oxide films (Al₂O₃ and complex oxides)
- Non-metallic inclusions
- Entrapped dross particles
Hydrogen solubility in molten aluminum is high at elevated temperatures but decreases sharply during solidification. This leads to gas pores forming inside castings if not properly removed.
In real production environments, insufficient melt treatment can increase defect rates by 10–30% depending on alloy and scrap ratio.
Refining flux is therefore not optional—it is a core part of melt quality control.

How Does Refining Flux Work in Molten Aluminum?
The effectiveness of refining flux comes from a combination of physical and chemical mechanisms rather than a single reaction.
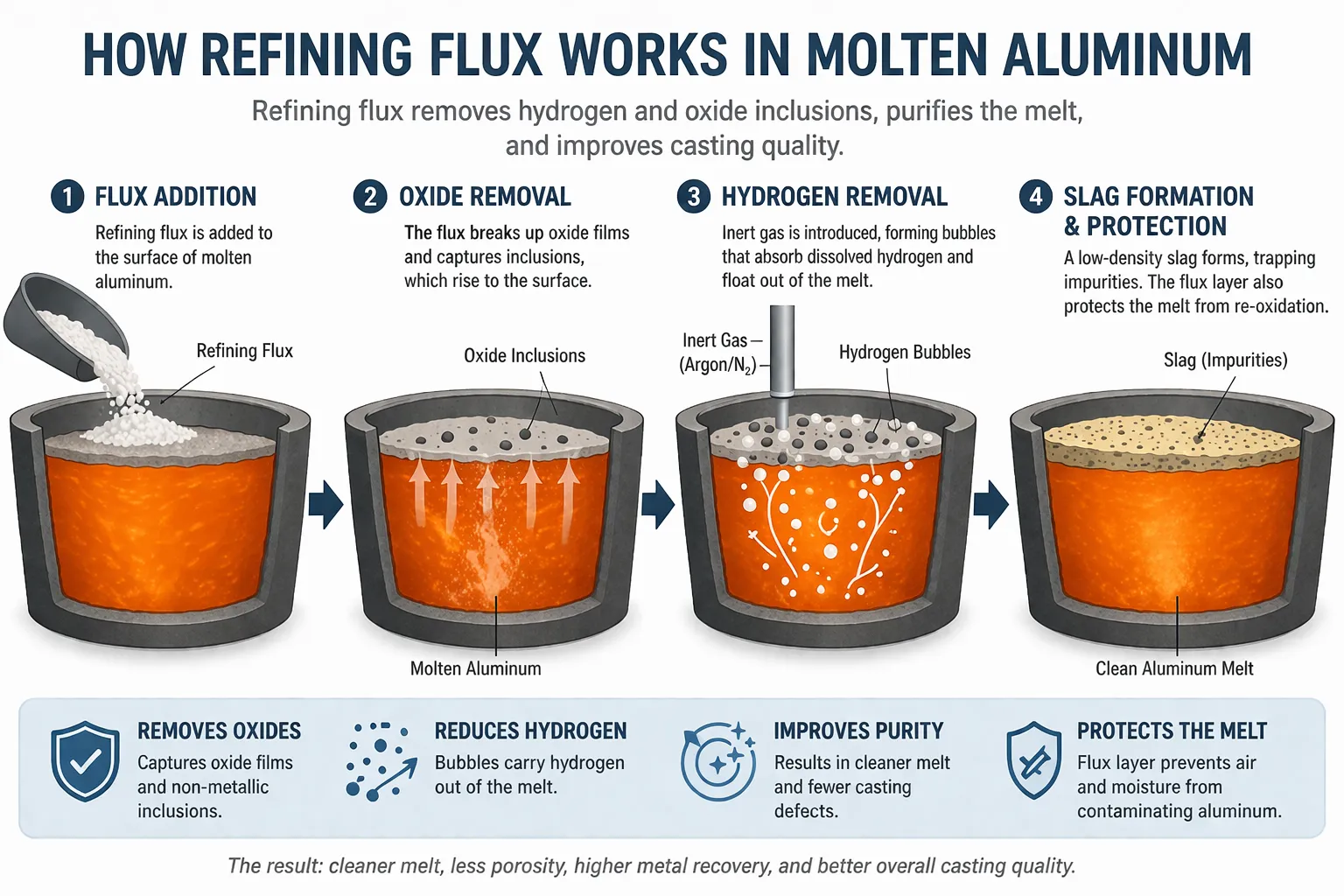
1. Interfacial Tension Reduction and Inclusion Removal
Refining flux lowers the surface tension between molten aluminum and oxide films. This allows trapped oxides to detach from the melt and rise to the surface.
2. Hydrogen Removal Through Bubble Transport
When used with inert gas injection (typically nitrogen or argon), flux improves bubble formation. Dissolved hydrogen diffuses into these bubbles and is carried out of the melt.
3. Slag Formation and Separation
Flux reacts with impurities to form a low-density slag layer that floats on the surface, making removal easier during skimming.
4. Melt Surface Protection
A flux layer reduces direct contact between molten aluminum and air, minimizing oxidation and preventing recontamination.
Practical observation from foundry operations:
Proper refining produces dry, powder-like dross with low metal loss, while poor treatment results in sticky slag containing trapped aluminum.
Looking for more guidance? Check out our “How to Use Aluminum Flux” guide or contact us.
What Is the Composition of Aluminum Refining Flux?
Most industrial refining fluxes are based on eutectic chloride systems with fluoride additives.
Table 1: Typical Composition of Refining Flux for Aluminum
| Component | Typical Range (%) | Function |
|---|---|---|
| Potassium Chloride (KCl) | 35–50 | Controls melting behavior and stability |
| Sodium Chloride (NaCl) | 35–50 | Lowers melting point and improves fluidity |
| Fluorides (NaF, CaF₂, Cryolite) | 2–10 | Break oxide films and enhance cleaning efficiency |
| Functional Additives | 0–10 | Improve reaction kinetics and flow behavior |
This composition reflects widely used industrial aluminum melt treatment formulations.
In practical application, fluoride activity is often the determining factor for oxide removal efficiency rather than base salt composition.
When Should Refining Flux Be Used in Aluminum Casting?
Timing directly affects performance. Even high-quality flux will underperform if used incorrectly.
Key Application Stages:
- After melting (initial cleaning stage)
- After alloying additions (composition stabilization)
- Before casting (final melt refinement)
Table 2: Flux Application Strategy in Industrial Production
| Stage | Purpose | Expected Result |
|---|---|---|
| Post-melting | Remove primary oxides | Cleaner baseline melt |
| After alloying | Remove reaction by-products | Stable chemical composition |
| Pre-casting | Final degassing and purification | Reduced porosity risk |
In high-scrap recycling environments, flux demand increases significantly due to higher oxide and contamination levels.
How Much Refining Flux for Aluminum Should Be Used?
Flux dosage depends on melt cleanliness, scrap ratio, and production requirements.
Table 3: Recommended Flux Addition Levels
| Melt Condition | Flux Addition (% of melt weight) |
|---|---|
| Clean primary aluminum | 0.2 – 0.5% |
| Standard alloy production | 0.5 – 1.0% |
| High scrap or contaminated melt | 1.0 – 2.0% |
Operational Best Practices:
- Add flux in multiple small doses rather than one large addition
- Ensure proper stirring or inert gas injection
- Maintain melt temperature between 700–750°C
- Allow sufficient reaction time (5–10 minutes minimum)
- Remove slag completely after treatment

Failure to follow these steps significantly reduces refining efficiency.
What Casting Defects Can Refining Flux Help Reduce?
Refining flux directly addresses several critical aluminum casting defects:
Gas Porosity
By removing dissolved hydrogen, flux reduces internal gas pore formation.
Oxide Inclusions
Flux captures oxide films and prevents them from remaining in the final casting.
High Metal Loss in Dross
Proper fluxing improves metal separation efficiency, reducing aluminum loss.
Inconsistent Mechanical Properties
Cleaner melts lead to more uniform microstructure and mechanical behavior.
In controlled production environments, proper flux use can reduce defect rates by 10–25%.
What Types of Aluminum Flux Are Used in Industry?
Different process requirements require different flux types:
- Oxide removal flux : break down and collect aluminum oxide inclusions formed during melting and handling
- Tundish flux :act as an insulating and protective layer on molten aluminum during transfer and holding
- Refining flux :remove dissolved hydrogen and fine inclusions
- Deslagging flux :separating aluminum from slag
Selecting the wrong type is one of the most common reasons for poor melt quality even when flux is used correctly.
Refining Flux vs Degassing vs Filtration: What Is the Difference?
Each melt treatment method targets a different problem.
| Method | Function | Limitation |
|---|---|---|
| Refining flux | Removes hydrogen and oxides | Requires correct process control |
| Degassing | Removes dissolved hydrogen | Does not remove oxides |
| Filtration | Removes solid inclusions | Does not remove dissolved gas |
Best Industrial Practice:
A combined system delivers the highest stability:
Refining Flux + Degassing + Filtration
This three-stage approach is widely used in automotive and aerospace casting production.
Common Mistakes When Using Refining Flux for Aluminum
Many performance issues are caused by incorrect application rather than the material itself.
1. Incorrect Temperature Control
Flux must fully melt and react. Low temperatures reduce effectiveness.
2. Insufficient Mixing
Without proper agitation, flux remains on the surface and does not treat the bulk melt.
3. Overuse of Flux
Excess flux increases slag volume and can introduce inclusions.
4. No Gas Injection Support
Hydrogen removal efficiency drops significantly without inert gas assistance.
5. Incomplete Slag Removal
Residual slag can reintroduce impurities into the melt.
Key insight:
Flux performance depends more on process discipline than product selection.
Real Foundry Experience: How Process Optimization Improved Melt Quality in Industrial Production
In real-world aluminum casting operations, one Southeast Asian aluminum foundry producing automotive components experienced persistent issues with gas porosity, unstable mechanical properties, and inconsistent surface quality.
Before optimization, the plant relied heavily on refining flux alone without a stable process control system. Although refining flux for aluminum was already in use, results remained inconsistent due to fluctuations in furnace temperature, irregular mixing practices, and unstable scrap ratios.
After implementing a structured melt treatment program—including optimized refining flux dosing, controlled nitrogen degassing, and standardized stirring procedures—under our technical guidance, the production line was significantly improved.
Key process adjustments included:
- Maintaining stable melt temperature between 700–750°C
- Applying refining flux in controlled multi-stage additions rather than single dosing
- Improving inert gas (argon/nitrogen) injection efficiency
- Standardizing holding time and slag removal procedures
- Strengthening scrap pre-treatment control
Within several production cycles, the foundry reported noticeable improvements in melt cleanliness and process stability. Defect rates related to porosity and oxide inclusions were significantly reduced, while dross metal loss was also lowered.
Most importantly, the plant achieved more consistent casting quality and higher production repeatability, allowing them to reduce rework and improve downstream machining efficiency.
The customer confirmed that the full performance of refining flux for aluminum can only be achieved when a high-quality flux is combined with correct process control and proper operational methods. In other words, optimal melt quality is the result of both effective refining flux and disciplined application practices working together.
FAQ
1. What is refining flux used for?
It removes hydrogen gas and non-metallic impurities from molten aluminum.
2. Does refining flux eliminate all casting defects?
No, but it significantly reduces porosity and inclusion-related defects.
3. What temperature is best for using flux?
Typically 700–750°C depending on alloy type.
4. Can flux remove porosity completely?
It reduces porosity but cannot eliminate all defects alone.
5. Is refining flux necessary for recycled aluminum?
Yes, scrap materials require more intensive melt treatment.
6. What gases are used with flux?
Nitrogen and argon are most commonly used.
7. Can too much flux cause problems?
Yes, overuse can increase inclusions and slag volume.
8. What industries use refining flux?
Automotive, aerospace, die casting, and aluminum recycling.
9. Is flux better than degassing?
No, both are complementary processes.
10. What indicates successful flux treatment?
Dry slag formation and reduced gas evolution.

