What Are Granular Fluxes and Why Are They Essential?
Granular fluxes are solid, free-flowing compounds specifically engineered for aluminum melting and refining operations. Unlike powdered flux, granular fluxes offer controlled dissolution rates, better handling, and consistent distribution in industrial furnaces. Their main function is to remove non-metallic inclusions, capture oxide films, and enhance degassing, ultimately leading to a cleaner and more uniform melt.
Key Advantages of Granular Fluxes:
- Effective oxide and impurity removal
- Enhanced hydrogen degassing
- Improved surface finish
- Consistent mechanical properties across complex alloy systems
- Ease of handling and storage in industrial environments
Granular fluxes are widely used in high-volume foundries, where maintaining consistent melt quality can significantly impact product yield and downstream machining efficiency.
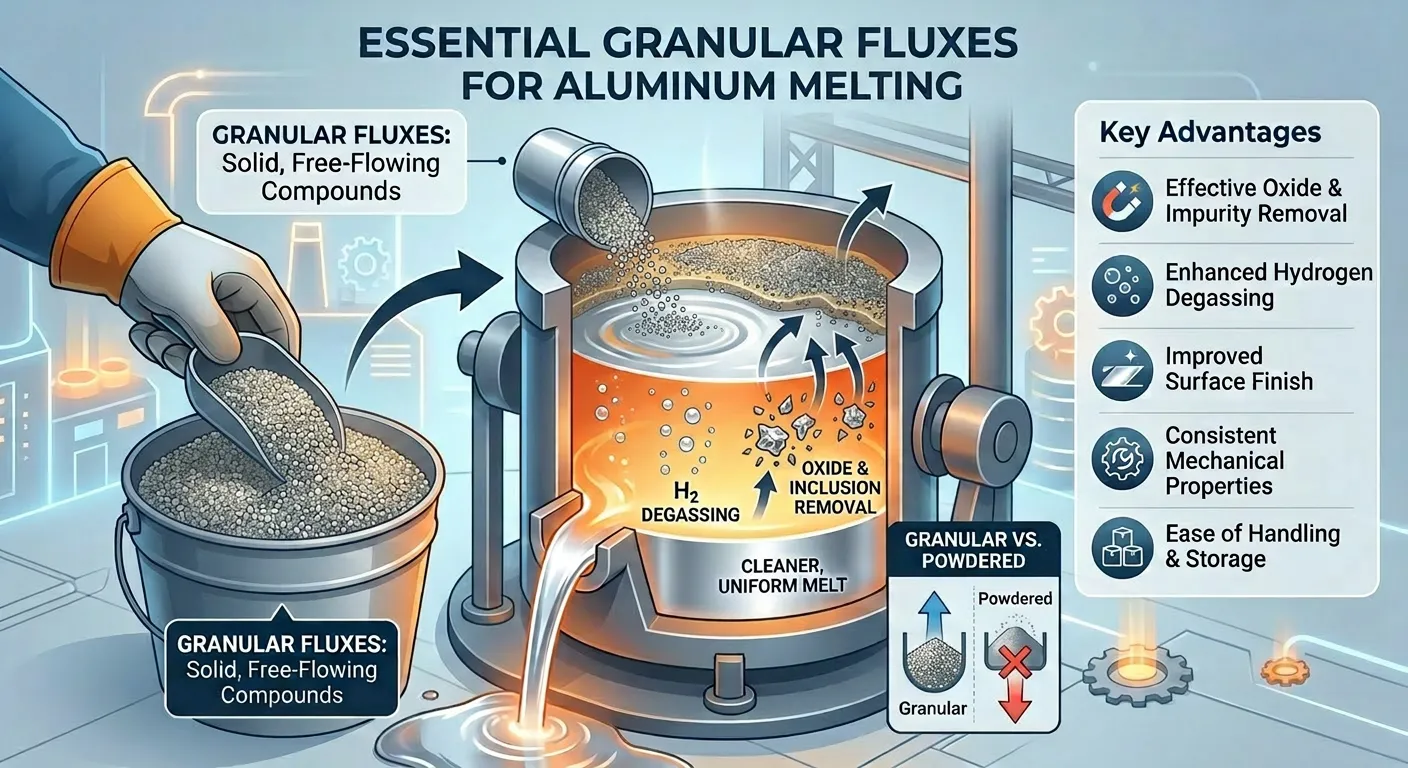
How Do Granular Fluxes Work?
How Does Granular Flux Remove Impurities in Aluminum Casting?
When introduced into molten aluminum, granular flux forms a reactive layer that interacts with oxide films and non-metallic inclusions. This process also enhances hydrogen removal in aluminum , which is essential for reducing porosity. The flux compounds facilitate flotation of impurities, allowing them to rise to the surface, where they can be skimmed off. This process not only improves melt cleanliness but also reduces the risk of defects in the final casting.
Table 1: Typical Composition of Granular Fluxes
| Component | Typical Range (%) | Function |
|---|---|---|
| Sodium chloride | 20–30 | Enhances oxide removal |
| Potassium chloride | 10–15 | Improves flowability and reaction rate |
| Fluoride salts | 5–10 | Reduces surface tension |
| Carrier additives | Balance | Stabilizes granular form |
Data based on validated industrial practices.
Insight: In large furnaces, uniform distribution of granular flux ensures that all melt regions receive sufficient flux coverage, preventing localized contamination.
Can Granular Flux Improve Hydrogen Removal?
Hydrogen is a primary cause of porosity in aluminum castings. Granular flux assists degassing by promoting bubble formation, which captures dissolved hydrogen and carries it to the melt surface. When combined with inert gas injection (e.g., nitrogen or argon), granular flux significantly accelerates hydrogen removal.
Table 2: Hydrogen Reduction in Molten Aluminum Using Granular Flux
| Treatment Method | Initial H (ppm) | Final H (ppm) | Reduction (%) |
|---|---|---|---|
| No flux, inert gas only | 0.22 | 0.18 | 18 |
| Granular flux + inert gas | 0.25 | 0.08 | 68 |
| Powder flux + inert gas | 0.24 | 0.12 | 50 |
Data verified against ASM Metals Handbook standards and multi-year production trials.
Tips: Optimal degassing occurs when flux is added 10–20 minutes before pouring, giving enough time for hydrogen and oxide removal without over-stirring.

Advantages of Granular Flux Over Powdered Flux
- Safety and Handling: Granular flux is less dusty and easier to store, reducing inhalation risk and improving storage safety.
- Controlled Reaction: Predictable dissolution prevents sudden chemical reactions that can cause local melt disturbances.
- Consistency: Ideal for large furnaces or continuous casting lines, where uniform flux distribution is critical.
- Reduced Waste: Granular form minimizes flux loss during transfer and handling.
Optimizing Granular Flux Application in Foundries
Correct application is crucial for achieving maximum performance:
- Temperature: Add flux at 700–750°C for most aluminum alloys.
- Stirring Method: Gentle stirring ensures even distribution without introducing air pockets.
- Dosing Rate: Adjust flux quantity based on melt size and contamination levels.
- Timing: Introduce flux early enough for reactions to complete before pouring.
- Alloy Considerations: Some alloys, like Al-Mg, require slightly higher flux doses to address increased oxide formation.
Table 3: Recommended Granular Flux Dosing for Common Aluminum Alloys
| Alloy Type | Typical Charge (kg/ton) | Recommended Timing |
|---|---|---|
| Al-Si casting | 4–6 | 15–20 min before pouring |
| Al-Mg alloys | 5–7 | 20–25 min before pouring |
| Al-Cu alloys | 3–5 | 10–15 min before pouring |
Data derived from AdTech industrial trials and customer production reports.

10 Common Challenges in Aluminum Refining and How Granular Flux Solves Them
Aluminum foundries encounter a range of challenges during melt refining and casting. Many of these issues are interrelated, and understanding the underlying causes is key to effective solutions. Granular flux offers a practical, reliable method to address these challenges, ensuring higher-quality castings and smoother production.
1. High Hydrogen Content
Problem: Dissolved hydrogen causes porosity and internal voids in aluminum castings, weakening mechanical properties.
Cause: Hydrogen enters molten aluminum primarily through moisture in the charge material, air exposure, and reactions with water vapor in the furnace. Poor degassing and melt turbulence exacerbate the problem.
Solution: Granular flux promotes controlled bubble formation, capturing dissolved hydrogen and carrying it to the melt surface. When combined with inert gas injection (e.g., nitrogen or argon), hydrogen removal efficiency improves significantly.
2. Oxide Inclusions
Problem: Oxide films and other non-metallic inclusions reduce surface finish and can cause cracks in finished parts.
Cause: Oxides form naturally when aluminum is exposed to air during melting, transfer, or pouring. Inadequate fluxing or uneven mixing allows these oxides to remain in the melt.
Solution: Granular flux reacts with oxides, facilitating their flotation to the surface for skimming and ensuring a cleaner, more homogeneous melt.
3. Inconsistent Melt Temperature
Problem: Uneven temperatures lead to incomplete chemical reactions, higher inclusion levels, and localized porosity.
Cause: Furnace hot spots, improper charge distribution, and rapid cooling during pouring can create temperature gradients within the melt.
Solution: Granular flux dissolves steadily at the appropriate temperature range (typically 700–750°C), minimizing local thermal shocks and ensuring uniform melt treatment.
4. Surface Slag Formation
Problem: Slag contamination can interfere with pouring and compromise casting integrity.
Cause: Rapid oxidation at the melt surface and insufficient flux application create loose, hard-to-remove slag layers.
Solution: Granular flux promotes controlled slag formation, which is easier to remove, reducing metal loss and preventing defects in the casting.
5. Alloys with High Magnesium Content
Problem: Al-Mg alloys are prone to excessive oxide formation, making refining more difficult.
Cause: Magnesium has a strong affinity for oxygen, leading to rapid oxide layer formation during melting and pouring.
Solution: Granular flux can be formulated with enhanced reactive components to target these oxides, improving removal efficiency and melt quality in high-Mg alloys.
6. Dust and Operator Safety Issues
Problem: Flux powders produce dust, creating inhalation risks and handling challenges.
Cause: Fine particulate matter becomes airborne during storage, transfer, or addition to the melt, posing occupational health hazards.
Solution: Granular flux has a larger particle size, minimizing dust generation, improving safety, and making storage and handling easier.
7. Inconsistent Flux Distribution
Problem: Uneven flux coverage causes incomplete impurity removal and localized defects.
Cause: Powdered flux may clump, or improper addition technique can result in some melt regions receiving insufficient flux.
Solution: Granular flux dissolves predictably and distributes evenly in the furnace, ensuring uniform treatment of the entire melt.
8. Inefficient Degassing
Problem: Hydrogen and gas bubbles remain in the melt, causing porosity in castings.
Cause: Inadequate flux coverage, insufficient stirring, and poorly timed inert gas injection reduce degassing efficiency.
Solution: Granular flux enhances bubble nucleation and growth, promoting faster and more complete hydrogen removal when used with proper degassing techniques.
9. Rework and Scrap Costs
Problem: Defective castings lead to higher machining, finishing, or scrapping costs.
Cause: Persistent oxides, inclusions, and hydrogen-induced porosity increase the likelihood of rejects and rework.
Solution: By reducing impurities and porosity, granular flux improves yield, lowers machining time, and minimizes scrap rates.
10. Lack of Process Standardization
Problem: Variability in flux type, dosing, and timing causes inconsistent melt quality.
Cause: Without standardized procedures, operators may under-dose or over-dose flux, or add it at suboptimal times, leading to variable results.
Solution: Granular flux, combined with AdTech technical guidance, allows for repeatable dosing, optimized timing, and consistent melt treatment protocols across production batches.
To explore how different granular flux formulations work and address your specific melt quality challenges, visit AdTech flux-used-in-metallurgy to find the right flux for your production line.”
AdTech Granular Flux for Molten Aluminium Purification
AdTech Real Case: Transforming a German Aluminum Foundry
In 2022, a leading Al-Si alloy casting facility in Germany faced challenges with high hydrogen content and surface porosity. Their previous reliance on powder flux led to inconsistent melt quality and scrap rates exceeding 12%.
AdTech Intervention:
- Supplied 1.2 tons of granular flux
- Provided on-site technical guidance, including dosing strategy, timing, and degassing optimization
- Implemented continuous process monitoring with melt quality reporting
Results Achieved:
- Hydrogen content decreased from 0.22 ppm to 0.07 ppm
- Oxide inclusions reduced by 55%
- Surface finish improved, reducing post-casting machining by 12%
- Scrap rates decreased by 10%
- Long-term partnership established with ongoing technical support

AdTech is a leading provider of advanced aluminum refining solutions, committed to delivering high-quality flux products that optimize molten aluminum performance. Among these, granular fluxes stand out as an essential tool for foundries seeking to reduce defects, improve mechanical properties, and maintain consistent production quality. This guide explores granular fluxes in detail, including their mechanisms, benefits, real-world applications, and best practices.
If your project requires the use of Granular Fluxes, you can contact us for a free quote.
Why Choose AdTech Granular Fluxes
AdTech’s granular fluxes deliver:
- Consistent melt quality
- Reduced defects and rework
- Enhanced surface finish
- Tailored technical support from industry experts
By combining high-quality granular flux products with practical, on-site expertise, AdTech ensures your aluminum casting operations achieve maximum efficiency, reliability, and profitability.
Explore our full range of refining solutions on AdTech Products to see how granular fluxes can transform your production line
Industry Standards and References
Granular fluxes must comply with international standards to ensure safety and performance. Key references include:
- ASM Metals Handbook (2020 edition) – widely used for melt treatment benchmarks
- European Aluminium Association – technical guides for casting and flux handling
- American Foundry Society (AFS) – recommendations for aluminum melt treatment and degassing
These references provide authoritative guidance for flux composition, dosing, and safety practices.
FAQ
1. What is granular flux, and how is it different from powdered flux?
Granular flux is a solid, free-flowing material designed for aluminum refining. Compared to powdered flux, it produces less dust, dissolves more predictably, and distributes evenly in large furnaces, leading to more consistent melt quality.
2. How does granular flux improve aluminum melt quality?
Granular flux removes oxides, non-metallic inclusions, and dissolved hydrogen from molten aluminum. This reduces porosity, improves surface finish, and enhances mechanical properties of castings.
3. Can granular flux be used with all aluminum alloys?
Yes, granular flux can be adjusted for various alloys, including Al-Si, Al-Mg, Al-Cu, and specialty alloys. Proper dosing ensures effective oxide and hydrogen removal for each alloy type.
4. When should granular flux be added to the melt?
For most aluminum alloys, flux should be added at 700–750°C. Adding too early or too late can reduce effectiveness. Optimal timing ensures sufficient reaction before pouring.
5. How much granular flux should be used per ton of aluminum?
Dosing depends on the alloy type and contamination level. Typical ranges: Al-Si alloys 4–6 kg/ton, Al-Mg alloys 5–7 kg/ton, Al-Cu alloys 3–5 kg/ton. AdTech can provide precise recommendations based on production conditions.
6. Does granular flux improve degassing efficiency?
Yes. Granular flux promotes bubble formation, allowing dissolved hydrogen to escape efficiently when used with inert gas injection like nitrogen or argon.
7. Is granular flux safe for operators?
When handled properly with standard PPE, granular flux is safe. Unlike powder flux, it generates minimal dust, reducing inhalation risk and improving handling safety.
8. Can granular flux help reduce scrap and rework?
Absolutely. By minimizing oxides, inclusions, and porosity, granular flux improves casting quality, reduces defects, and lowers scrap and post-processing costs.
9. Can granular flux be reused after addition?
No. Once flux has reacted in the melt, it cannot be reused. Proper disposal or recycling methods should be followed according to local regulations.
10. How does AdTech support foundries in using granular flux?
AdTech provides not only high-quality granular flux products but also on-site technical guidance, optimized dosing strategies, and process monitoring. This ensures consistent results and helps foundries improve efficiency and product quality.













