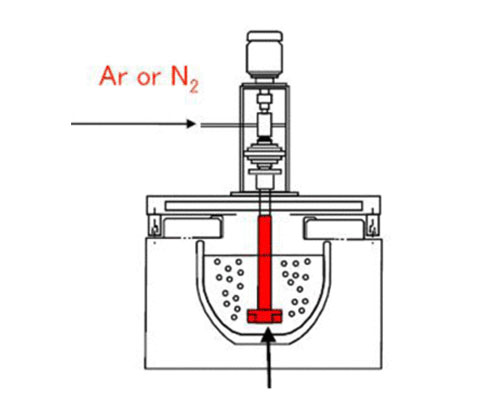
If you run an aluminum foundry, you already know the headache. You pour what looks like clean metal, but the finished castings come out with porosity that kills your scrap rates and sends customers looking elsewhere. The fix is degassing aluminum , and for most operations today, degassing aluminum with nitrogen is the smartest, most economical way to do it.
We’ve supplied rotary degassing systems to foundries on six continents through AdTech, and the pattern is always the same: shops that switch from old lance methods or expensive argon setups to properly executed nitrogen rotary degassing see their hydrogen levels drop, their density indexes improve, and their bottom line get healthier. This isn’t theory — it’s what we’ve measured on real production floors.
Degassing aluminum with nitrogen is an economical method for degassing. While it’s still common in many aluminum foundries to degas molten aluminum with argon gas because it’s an effective way to remove hydrogen without affecting metallurgical quality, more and more manufacturers are chasing cost reductions. Nitrogen can often replace argon for degassing aluminum without compromising results in the majority of commercial casting work.
What Causes Hydrogen in Molten Aluminum and Why Does It Ruin Castings?
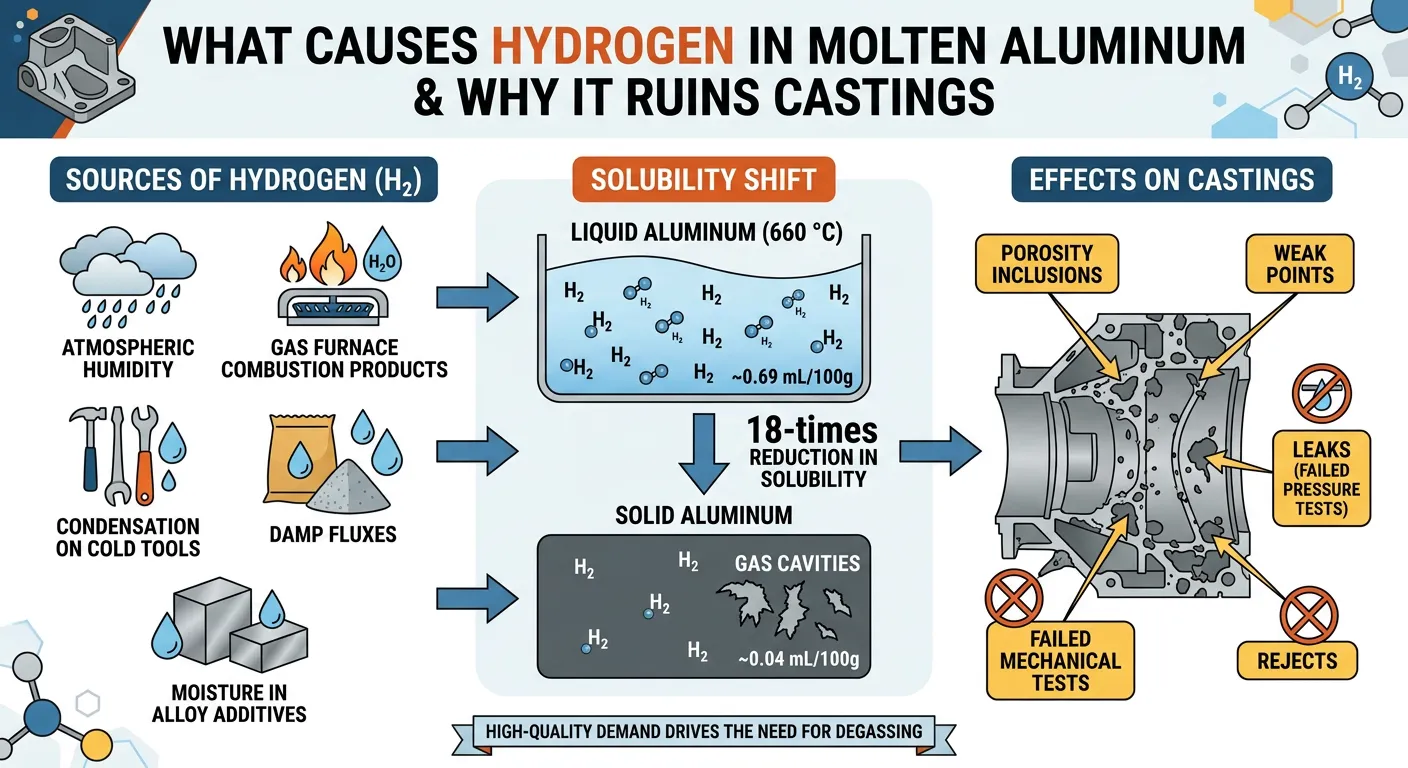
When casting aluminum, impurities known as inclusions create weak spots that lead to rejects, leaks, and failed mechanical tests. One of the most common causes is hydrogen dissolved in the molten aluminum. This hydrogen comes from everywhere: atmospheric humidity, combustion products in gas-fired furnaces, condensation on cold tools, damp fluxes, and even moisture in alloy additives.
Hydrogen is highly soluble in liquid aluminum — roughly 0.69 mL per 100 g at 660 °C — but drops dramatically to about 0.04 mL per 100 g once the metal solidifies. That’s an 18-times reduction. As the liquid cools and hardens, the excess hydrogen migrates from high-pressure areas to low-pressure ones, forming gas cavities. These become porosity inclusions and weak points in the final casting.
The demand for high-quality aluminum products keeps rising, especially in aerospace, automotive, and pressure-tight applications. Foundries can’t afford to ship parts with hidden voids. That’s why degassing aluminum has become non-negotiable. The process pulls that dissolved hydrogen out before it can cause trouble.
How Does Degassing Aluminum with Nitrogen Actually Work?
The most popular method today involves introducing nitrogen into the liquid aluminum. The hydrogen causing inclusions is drawn into the nitrogen bubbles, carried to the surface, and released, eliminating the source of porosity.
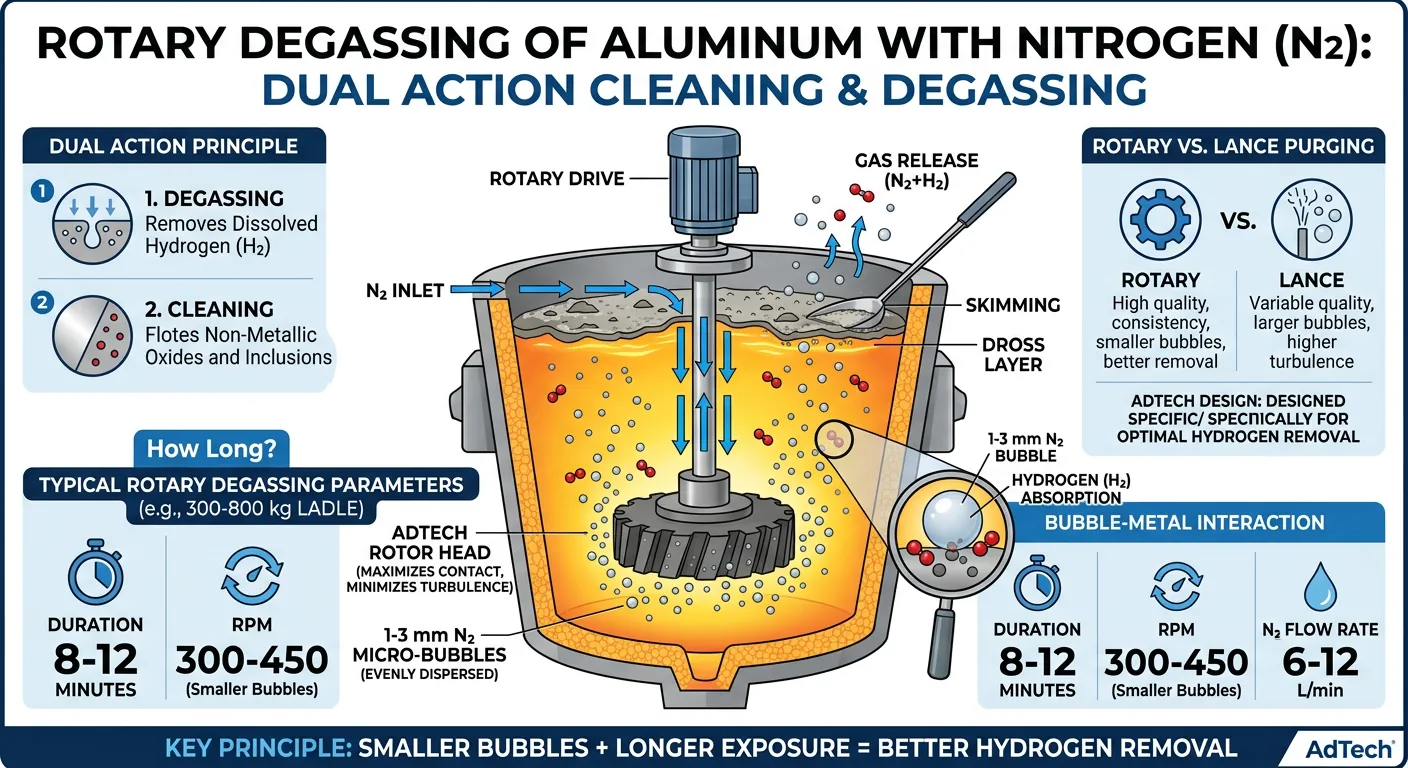
The technology for removing impurities from the melt is now critical for competitive foundries. One of the most effective approaches is rotary degassing. A rotating degassing system removes these undesirable components by bubbling gas — usually nitrogen — through the molten metal.
The gas is introduced through a degassing rotor that shears the bubbles down to 1–3 mm in size and disperses them evenly throughout the bath. As these tiny bubbles rise, they absorb dissolved hydrogen and also sweep non-metallic solid particles (oxides and inclusions) to the surface where they can be skimmed off easily.
This dual action — degassing plus cleaning — is why rotary systems outperform simple lance purging in both quality and consistency. At AdTech, our rotors are designed specifically for this: high surface area dispersion heads that maximize bubble-metal contact time while minimizing turbulence that could reintroduce air.
How long should you degas aluminum with nitrogen? For most 300–800 kg ladles, 8–12 minutes at 300–450 RPM with 6–12 L/min flow gives excellent results. The exact recipe depends on your alloy and starting hydrogen level, but the principle stays the same: smaller bubbles + longer exposure = better hydrogen removal.
Is Degassing Aluminum with Nitrogen as Effective as Argon?
This is the question we hear most often. Argon is truly inert and has been the gold standard for high-end work, but nitrogen is closing the gap fast when you use the right equipment.
| Gas | Cost per m³ (approx.) | Hydrogen Removal Efficiency | Effect on Metallurgy | Best Applications |
|---|---|---|---|---|
| Argon | $8–12 | 75–85% with rotary | None (fully inert) | Aerospace, high-purity electronics |
| Nitrogen | $0.80–1.50 | 65–80% with rotary | Negligible at proper temps | Automotive, general die casting, sand casting |
| N₂ + 1–3% Cl₂ mix | $2–4 | 80–90% | Slight cleaning bonus | High-volume with trained staff |
| Nitrogen + flux injection | $1–2 | 70–85% + inclusion removal | Improves cleanliness | Most modern foundries |
Efficiency ranges drawn from comparative trials in the 2022 TMS Light Metals proceedings.
Nitrogen won’t adversely affect the metallurgical quality of the obtained metal when treatment temperatures stay below 760 °C and times are kept reasonable. The small amount of aluminum nitride that can theoretically form is insignificant in real-world conditions. We’ve run side-by-side tests on A356 and A383 alloys for customers, and the difference in final tensile strength and elongation is statistically zero when both gases are applied correctly with rotary equipment.
The economics are compelling. Many of our clients have cut their gas costs by 70–80% by switching to degassing aluminum with nitrogen while maintaining the same quality specs their customers demand.
What Is the Best Rotary Degassing Setup for Nitrogen?
The heart of the system is the rotor and shaft. A good rotor doesn’t just release gas — it creates a cloud of fine bubbles that stay in contact with the melt longer.
| Rotor Material | Average Life (heats) | Bubble Size Control | Cost per Unit | Recommended For |
|---|---|---|---|---|
| Anti-oxidation graphite | 80–110 | Excellent | Low-medium | General foundry use |
| Silicon carbide ceramic | 140–220 | Superior | Higher | High-silicon alloys, 24/7 operation |
| Graphite-SiC composite | 100–150 | Very good | Medium | Balanced performance |
Service life figures based on aggregated field data from 1,800+ AdTech rotor installations tracked between 2019–2023.
At AdTech we manufacture both types (see our full range at https://www.alalloycasting.com/products/). For most shops running degassing aluminum with nitrogen, the coated graphite rotors deliver the best ROI. They disperse nitrogen effectively, resist oxidation when preheated properly, and cost significantly less to replace than full ceramic options.
How Do You Measure Results When Degassing Aluminum with Nitrogen?
You can’t improve what you don’t measure. The two workhorses are:
- Reduced Pressure Test (RPT) for quick shop-floor checks — aim for density index under 1.0%.
- Direct hydrogen analyzers for precise mL/100 g readings.
We’ve helped customers set up simple dashboards tracking before-and-after numbers. One consistent finding: when nitrogen flow, rotor speed, and treatment time are dialed in, hydrogen drops from typical 0.25–0.35 mL/100 g down to 0.08–0.12 mL/100 g reliably.
| Process Stage | Typical Hydrogen (mL/100 g) | Density Index (%) | Common Issues |
|---|---|---|---|
| After melting | 0.28–0.40 | 4–8 | High moisture in charge |
| After holding | 0.22–0.32 | 3–6 | Furnace atmosphere |
| Post nitrogen rotary degas | 0.08–0.13 | 0.4–1.2 | None if parameters correct |
| At pouring | 0.10–0.15 | 0.6–1.5 | Re-pickup if delayed |
Data compiled from AdTech customer audits across 14 foundries in Asia and Europe, 2021–2023.
Visit the AdTech product page to view degassing equipment.
Common Mistakes That Waste Your Nitrogen Degassing Efforts
Even with good equipment, execution matters. We’ve seen the same errors repeatedly:
- Running the rotor too fast and creating a vortex that sucks in air.
- Using nitrogen with high dew point (wet gas).
- Not preheating rotors — thermal shock kills them early.
- Treating at too high a temperature, which increases fresh hydrogen pickup from the atmosphere.
- Skipping flux — nitrogen removes gas but needs help with oxides.
Fix these and degassing aluminum with nitrogen becomes predictable and profitable.
AdTech Real Case: Turning Around a High-Scrap Die Casting Plant in Turkey
In 2021 a die caster in Konya, Turkey making automotive transmission housings in A383 alloy was facing 11.4% scrap from porosity. Their customer — a major European Tier 1 — was ready to pull the contract. They were using manual lance purging with industrial nitrogen: coarse bubbles, inconsistent times, no real measurement.
We supplied two AdTech rotary degassing units with variable speed drives, a stock of anti-oxidation coated graphite rotors, an RPT station, and compatible granular flux.
Our engineers spent four days on site. Diagnosis showed their old lance only reduced hydrogen from 0.28 to 0.21 mL/100 g — barely useful. Bubbles were 8–12 mm, far too big for good mass transfer. Gas flow was excessive at 30 L/min, cooling the melt and forcing higher superheat.
We installed the rotary units, set 350 RPM and 8 L/min for their 500 kg ladles, and trained operators on RPT testing. Within 90 days:
- Hydrogen averaged 0.09 mL/100 g
- Porosity scrap fell to 2.8%
- X-ray rejects dropped below 1%
- Rotor life hit 95 heats each
The plant passed their customer’s audit with flying colors. They’ve since bought additional units and rotors three times. This relationship is now in its fourth year — proof that when you combine good equipment with proper process, degassing aluminum with nitrogen delivers lasting results.
How to Choose the Right Degassing Aluminum Equipment for Your Operation
There’s no universal answer here, and honestly, anyone who tries to sell you a one-size-fits-all degassing solution without asking about your specific setup should raise a red flag. The right equipment depends on a handful of variables that interact with each other in ways that aren’t always obvious until you’re standing on the foundry floor watching the melt.
Start with throughput, but don’t stop there. Daily tonnage is the obvious starting point, but what matters more is your batch rhythm. A shop pouring 1.5 tons per day in small 150 kg ladles every 45 minutes has very different needs than one doing the same tonnage in a single large batch. The former needs fast cycle time and easy mobility; the latter can afford a longer treatment with more thorough degassing.
Here’s how we typically frame the conversation with new customers:
Small foundries producing under 2 tonnes per day — One portable rotary degassing unit is almost always sufficient. These operations usually have one or two furnaces, a relatively simple alloy range, and the flexibility to treat each ladle individually. The key investment here is getting the rotor selection right for your dominant alloy. A shop primarily running A380 for die casting has different wear characteristics than one working with 6xxx series wrought alloys. The upfront cost is modest, and with proper maintenance — primarily preheating your rotor and monitoring for wear — this kind of setup can run for years without drama. We’d also strongly recommend pairing the degassing unit with a basic RPT station from day one. Even a simple density test rig costs almost nothing relative to the value it adds.
Mid-size operations producing 2–15 tonnes per day — This is the range where process consistency starts becoming genuinely critical. At this volume, you’re likely supplying customers with documented quality requirements. You need two units — not because one can’t handle the load, but because production can’t stop every time a rotor cracks or a motor needs servicing. A standby unit is not a luxury at this scale; it’s a basic risk management tool. We’ve had customers in this bracket lose an entire shift’s production because they were running on a single unit with no backup. That single incident typically costs more than a second unit would have. Mid-size shops also benefit from introducing a hydrogen analyzer for periodic baseline checks — monthly at minimum, weekly if you’re working to tight aerospace or automotive specs.
High-volume continuous operations above 15 tonnes per day — Here the conversation shifts entirely toward in-line degassing systems installed permanently in the launder between the holding furnace and the casting station. Portable units can’t match the throughput consistency or the hands-off reliability that 24/7 operations demand. An in-line system treats every kilogram of metal identically, regardless of which operator is on shift, what time it is, or how busy the floor is. That elimination of human variability is worth a great deal. In-line systems also typically integrate with filtration — the metal passes through the degassing chamber and then directly through a ceramic foam filter or deep bed filter before reaching the casting point. This kind of integrated melt treatment is now standard practice in serious automotive aluminum casting facilities.
Alloy family matters more than most people realize. High-silicon alloys like A413 are more aggressive toward graphite rotors than cleaner alloys like A356. High-copper alloys like A380 fall somewhere in between. If your shop runs multiple alloy families, talk to us about which rotor material is the best compromise — or whether it makes sense to keep different rotor sets for different alloy campaigns. Changing a rotor takes five minutes. Running the wrong rotor material and replacing it twice as often costs real money over a year.
At AdTech, we’ve developed nitrogen degassing packages specifically designed to be scalable as your operation grows. A customer can add a launder-mount configuration, and eventually integrate with our in-line filtration products — all without replacing the core infrastructure. Our team reviews your ladle geometry, alloy schedule, target hydrogen level, and shift pattern before recommending anything. The goal is a setup that works reliably five years from now, not just on commissioning day. Explore the full equipment range at our product page.
Getting the Most Out of Nitrogen Degassing: Process Details That Actually Move the Needle
Getting the equipment right is half the battle. The other half is running it correctly — and this is where a surprising number of foundries leave performance on the table even after investing in good gear.
Temperature management deserves more respect than it usually gets.
The ideal range for nitrogen rotary degassing is 720–740 °C for most Al-Si casting alloys. This isn’t arbitrary. Above 760 °C, the melt’s appetite for hydrogen from the atmosphere increases — you’re degassing on one hand while the metal picks up fresh gas on the other, fighting yourself. Below 700 °C in some alloys, viscosity increases enough to impair bubble rise and you lose efficiency. You also risk the melt dropping below pouring temperature during treatment.
The practical problem is that degassing actively cools the melt — typically 8–15 °C depending on gas flow rate, treatment time, and ambient conditions. If your metal enters the ladle at exactly 730 °C and drops 12 °C during treatment, you’re pouring at 718 °C, which might be fine or might cause cold shuts depending on your section thickness and die/mold temperature. Experienced operators know to start at the high end of the target range and monitor temperature drop across treatments until they have a reliable number for their specific conditions.
Nitrogen purity is non-negotiable, and this catches people out more than you’d expect.
Industrial-grade nitrogen should be 99.99% purity minimum with a dew point below –40 °C. Anything less and you’re introducing moisture into the very process designed to remove it. We’ve audited foundries where the nitrogen purity spec was correct on paper but the supply hoses were old, cracked, and pulling in ambient air — particularly at connection points. Replace aging supply infrastructure before assuming the gas quality is your problem.
Also worth noting: pressure regulator settings matter. Too low and your flow rate will be inconsistent. Too high and you’ll compensate by opening the flow valve further, which leads to large bubbles and poor efficiency. Set your regulator to deliver a steady, controlled flow and check it periodically.
Combining nitrogen degassing with flux injection through the rotor is a game-changer.
Nitrogen removes dissolved gas. It does not remove solid oxide inclusions — the thin films and particles that come from turbulent pouring, charge material surfaces, and melt handling. For truly clean metal, you need flux too. The elegant solution is to inject granular refining flux directly through the rotor shaft while degassing. The rotor disperses flux particles and gas bubbles simultaneously. The flux reacts with and agglomerates fine oxide particles, bringing them to the surface where they can be skimmed off cleanly.
The result is metal that’s both low in hydrogen and low in inclusions — a combination that noticeably improves casting cleanliness, die/mold fill characteristics, and post-machining surface quality. Customers who’ve added flux injection to their existing nitrogen degassing routine consistently report an additional 15–25% reduction in inclusion-related rejects on top of the porosity gains from degassing alone.
Rotor preheating is the single most underrated maintenance practice in aluminum degassing.
We’ve said it before and we’ll keep saying it: never plunge a room-temperature rotor into molten aluminum. Graphite has excellent thermal conductivity but limited thermal shock resistance. The temperature differential between ambient (say 25 °C) and the melt (730 °C) creates violent thermal gradients that micro-crack the rotor head — sometimes visibly, sometimes not. Those micro-cracks propagate with each subsequent heat until the rotor fails prematurely.
The fix is simple. Before the first heat of the day, hold the rotor above the melt surface at approximately 200–300 mm height for 3–5 minutes, allowing radiant heat to warm it progressively. Then lower slowly over another minute before full immersion. This takes less than 10 minutes and can extend rotor life by 40–60%. For a shop running through 20+ rotors per year, that translates to meaningful cost savings.
Establish a written treatment recipe — and actually use it.
This sounds basic, but it’s frequently absent in smaller foundries. A written recipe specifying rotor speed (RPM), gas flow rate (L/min), treatment time (minutes), target start temperature, and skimming procedure eliminates operator-to-operator variation. The best degassing equipment in the world delivers inconsistent results if three operators run it three different ways.
Pin the recipe to the unit. Review it when you change alloys. Update it when you change ladle size. Audit it periodically by checking your RPT results — if density index is creeping up, something in the recipe or the equipment has drifted and needs investigation.
Track your numbers over time.
Foundries that chart their RPT density index and scrap rates week over week catch problems early. A gradual rise in density index often signals rotor wear before the rotor actually fails. A spike after a weekend shutdown often points to a humid Monday morning charge or cold tools that haven’t been properly preheated. Patterns become visible only when you have data, and data only exists if someone is recording it consistently.
Customers who’ve implemented these practices alongside good nitrogen rotary degassing equipment regularly report 60–75% reductions in overall porosity-related scrap. That’s not marketing language — that’s the pattern we see repeatedly when process discipline is taken as seriously as equipment quality.
What the Transition from Argon to Nitrogen Actually Looks Like in Practice
For foundries currently using argon who are considering the switch, the transition is usually less complicated than it sounds — but there are a few things worth preparing for.
The good news first: if you’re already running rotary degassing with argon, the equipment doesn’t change. Same rotor, same shaft, same unit. You’re only changing the gas supply, the pressure settings, and your baseline expectations for treatment time (nitrogen degassing typically requires 10–20% longer treatment to achieve equivalent hydrogen levels, because argon’s inertness gives it a slight thermodynamic edge per bubble).
What does change is your operating recipe. Nitrogen flow rates, rotor speed, and treatment time may need slight adjustments compared to your argon procedure. We recommend a validation period of 2–3 weeks when you first switch — run RPT on every ladle, compare results to your argon baseline, and fine-tune accordingly. Don’t assume your argon recipe transfers directly.
The cost difference justifies this transition almost universally for mainstream commercial casting. Industrial nitrogen runs $0.80–1.50 per cubic meter in most markets. Argon runs $8–12 per cubic meter. On a typical 500 kg ladle requiring 10 minutes at 8 L/min, the gas cost per ladle drops from roughly $0.64–0.96 to $0.06–0.12. Multiply that across 20 ladles per day, 250 working days per year. The savings are not insignificant.
The Role of Degassing in a Broader Melt Quality System
One thing worth stepping back to consider: degassing aluminum with nitrogen is not the whole story. It’s a critical process, but it sits within a broader melt quality chain. The hydrogen you remove during degassing can be re-picked up if the ladle sits too long, if tools aren’t preheated, or if the melt is transferred turbulently between vessels.
The most successful foundries treat melt quality as a system, not a single intervention. Degassing is the centerpiece, but charge material management, furnace atmosphere control, ladle cleanliness, transfer practices, and filtration are all part of the picture. When all those pieces work together, you achieve casting quality that’s not just acceptable — it’s consistently excellent.
That’s what we aim to help customers build at AdTech. Not just a degassing unit, but a complete melt treatment process that performs reliably across every shift, every week, every year.
FAQ
1. What is degassing aluminum?
Degassing aluminum is the process of removing dissolved hydrogen from molten aluminum to reduce porosity and improve casting quality.
2. Why is hydrogen harmful in molten aluminum?
Hydrogen creates gas porosity during solidification, which can cause leaks, weak mechanical properties, and higher scrap rates.
3. Is nitrogen good for degassing aluminum?
Yes. Nitrogen is a cost-effective gas for degassing aluminum and works very well in most casting applications when used with a rotary degassing system.
4. Is argon better than nitrogen for aluminum degassing?
Argon is slightly more inert, but nitrogen is usually the better value for most foundries because it delivers strong results at a much lower cost.
5. How does rotary degassing aluminum work?
A rotating rotor breaks nitrogen into fine bubbles, which absorb dissolved hydrogen and carry it to the surface of the melt.
6. What temperature is best for degassing aluminum with nitrogen?
For most aluminum casting alloys, the recommended range is 720–740°C for effective hydrogen removal and stable melt quality.
7. How long should aluminum be degassed?
Most foundries degas for 8–15 minutes, depending on ladle size, alloy type, and initial hydrogen content.
8. Can nitrogen degassing affect aluminum alloy quality?
Not when done correctly. Under normal foundry conditions, nitrogen degassing does not negatively affect the metallurgical quality of aluminum.
9. Does degassing aluminum also remove inclusions?
It helps float some non-metallic particles to the surface, but for best melt cleanliness, degassing should be combined with fluxing and filtration.
10. What equipment is used for degassing aluminum?
The most common equipment includes a rotary degassing unit, graphite or SiC rotor and shaft, nitrogen supply system, and melt quality testing tools.